Anogenital warts
Peer reviewed by Dr Colin Tidy, MRCGPLast updated by Dr Hayley Willacy, FRCGP Last updated 27 Oct 2021
Meets Patient’s editorial guidelines
Medical Professionals
Professional Reference articles are designed for health professionals to use. They are written by UK doctors and based on research evidence, UK and European Guidelines. You may find the Genital warts article more useful, or one of our other health articles.
In this article:
Synonyms: human papillomavirus (HPV), condylomata acuminata, condyloma acuminata, genital warts, penile warts, vulval warts, labial warts, anogenital warts, vaginal warts, cervical warts
Continue reading below
What are anogenital warts?
Anogenital warts are benign proliferative epithelial growths caused by infection with human papillomavirus (HPV). They are very common, particularly in young people. However, it is hoped incidence will decline significantly as a result of widespread HPV vaccination. All those with anogenital warts should be referred to specialist sexual health clinics for assessment and treatment.
Anogenital warts causes1 2
Anogenital warts are caused by infection with HPV. There are >100 of these double-stranded-DNA papovaviruses characterised, with most now fully DNA-sequenced.
HPV is transmitted sexually in most cases but can also be transmitted prenatally, by auto-inoculation or hetero-inoculation from non-genital warts.
Anogenital warts have a 60% transmission rate between partners.
Over 90% of genital warts are caused by infection with HPV types 6 and 11. These types are not associated with a significant risk of neoplastic transformation.
Types 16 and 18 are associated with a high risk of neoplastic transformation. There may be co-infection with these high-risk types of HPV.
Risk factors for anogenital warts3
Smoking.
Multiple sexual partners.
Early age of onset of sexual intercourse.
History of other sexually transmitted infections (STIs). (Up to 20% of those with anogenital warts have other STIs.)
Anoreceptive intercourse. However, perianal warts can occur in the absence of anal intercourse. Perianal warts occur in men and women but are most common in men who have sex with men2 . Warts within the anal canal are usually, but not always, associated with penetrative anal intercourse1 .
Manual sexual practices such as fisting and fingering may increase the risk of anal warts4 .
Immunosuppression (including iatrogenic variety in transplant recipients and HIV)5 .
Continue reading below
Epidemiology
The estimated annual incidence of genital warts in developed world populations was about 0.15% of the adult population per year2 . The quadrivalent vaccine (HPV 6/11/16/18) is intended to provide protection against the principal causes of both benign and malignant genital HPV disease and has been used in the HPV vaccine programme in the UK since 2012. Genital wart rates are expected to fall, at least in vaccinated cohorts, as has been reported elsewhere - eg, Australia6 . Genital warts remain, however, one of the most common conditions treated in GUM clinics, with over 130,000 cases treated a year in the UK.
Anogenital warts symptoms1 2
Usually the presentation is with the presence of a lesion or lesions, which are usually painless. Lesions may be disfiguring or embarrassing. They may cause itching, bleeding or dyspareunia. Urethral lesions may cause distortion of the urinary stream.
Female anogenital warts
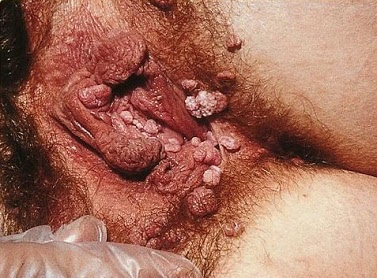
By SOA-AIDS Amsterdam, via Wikimedia Commons
A relevant sexual history should be obtained to assess the risk of other STIs and sexual health needs7 :
Other symptoms - eg, urethral/vaginal discharge, pelvic or scrotal pain, intermenstrual or postcoital bleeding, dyspareunia.
Sexual activity in the preceding three months.
Contraceptive and condom use.
Possibility of pregnancy.
HIV risk activities.
Signs2
Lesions may be single but are often multiple. A number of lesions may form a confluent mass. They may become huge in immunocompromised patients.
Warts on moist, non-hairy skin are usually soft and non-keratinised, whereas those on dry hairy skin are more likely to be firm and keratinised.
Warts may be broad-based or pedunculated.
Warts may be pigmented or not. Colour varies and may be pink, red, white, grey or brown.
They are often found on areas subject to trauma during sexual intercourse.
Sites where lesions are found include:
In uncircumcised males: frenulum, corona, glans penis inner foreskin, as well as the urethral meatus, penile shaft and scrotum.
In circumcised males: mainly the penile shaft, also the urethral meatus and scrotum.
In females: labia, clitoris, urethral meatus, introitus, vagina and cervix.
Both sexes: perineum, groin, pubis, perineum, perianal area and anal canal.
The anogenital and surrounding skin should be examined under good illumination. Female patients should undergo a vaginal speculum examination and the magnification provided by colposcopy may be useful. Proctoscopy may be indicated in both sexes if there is a history of anoreceptive sex or in patients with warts at the anal margin where the upper limit cannot be visualised, or in those with other anal canal symptoms such as irritation, bleeding or discharge. Meatoscopy should be performed if there is difficulty in visualising the full extent of intra-meatal warts. Occasionally urethroscopy is indicated for more proximal warts.
Recording lesions on genital maps can be useful to enable a visual record and monitor response to treatment.
Continue reading below
Investigation2
Diagnosis by biopsy and viral typing is not routinely required and tends to be reserved for where diagnosis is uncertain or for recalcitrant warts, warts with atypical features or where there is high risk of HPV-related malignancy.
Be suspicious of unusual presentations (eg, flat or only slightly raised lesions, pigmented, indurated or ulcerated warts), particularly in patients aged over 35 years or with other risk factors. Also be aware of the development of new symptoms (eg, itching, pain, crusting, bleeding) and warts that are unresponsive to treatment. Arrange for biopsy to exclude malignancy in these situations.
An appropriate screen for other STIs should be carried out.
Differential diagnosis8
Hair follicles.
Sebaceous glands.
Skin tags.
Pearly penile papules (normal variant: 1-3 rows of smooth, discrete, non-coalescing, 1-2 mm papules appearing on the margin of the glans)9 .
Vulval papillomatosis (normal variant: regularly shaped, non-coalescing, largely symmetrical papillae on the inside of the labia minora and vestibule)10 .
Hymen remnants.
Intra-epithelial neoplasia (vulval, anal or penile).
Malignancy.
Condyloma lata (secondary syphilis).
Associated diseases
Up to 20% of those with anogenital warts will have another STI.
The majority of external genital warts are caused by HPV types 6 and 11 which are not associated with significantly increased risk of neoplastic transformation. However, a minority of genital warts are caused by HPV types 16 and 18 and there is also a significant risk of co-infection with these types. These oncogenic types are associated with an increased risk of11 :
Anogenital warts treatment2 7
General points
Patients will require a detailed explanation of the condition with emphasis on long-term health implications for themselves and their partners. Reinforce with written information. Explain the long latent period associated with HPV and that recurrence of warts in one partner does not imply infidelity.
Data are conflicting regarding condom use. Consistent condom use has been shown to reduce the risk of acquisition of genital warts (in the order of 30-60% reduction) in some studies but that it may not protect against transmission of HPV in others. Condom use is advised until resolution of lesions has occurred. NB: latex condoms may be weakened if in contact with imiquimod.
Individuals should be aware that the HPV persists after clinical clearance of warts for very variable lengths of time.
Psychological distress is common - referral for counselling may be appropriate.
20% or more of patients with genital warts have concurrent STIs, so appropriate screening should be discussed and offered (including chlamydia, gonorrhoea, HIV, syphilis and hepatitis B and C).
Current partners and if possible partners in the previous six months should be assessed and educated with regard to prevention of STIs.
Advice about smoking cessation should be given. There is evidence that non-smokers have a better response to treatment than smokers.
Referral
Ideally anyone with anogenital warts should be referred to a sexual health clinic. This is because in the specialist setting:
There is more accurate testing for other STIs and there are better facilities for treating and contact tracing where associated infections are found.
There is better diagnostic accuracy.
A full range of treatment options is available, including surgical ablation.
Consistent information is available.
There is additional confidentiality.
Referral is particularly important where:
The diagnosis is unclear.
There is suspicion of intraepithelial neoplasia or malignancy (urgent referral).
There are internal warts in difficult-to-reach sites (intravaginal, cervical, urethral meatus or intra-anal).
The patient is a child, pregnant or immunosuppressed.
There is treatment failure or where treatment cannot be tolerated due to side-effects.
There are problematic recurrences.
There are positive results from the screen for other STIs.
Treatment options2 7
No treatment. 30% of warts regress spontaneously within six months.
The evidence base to direct first-line and second-line treatments is not strong and all treatments have significant failure and relapse rates.
Podophyllotoxin self-application is suitable for soft, non-keratinised external genital warts. 0.15% cream or 0.5% solution should be used twice-daily for three days, followed by four rest days. If the wart persists, treatment may be repeated at weekly intervals, for a total of five weeks. The solution is more convenient for penile warts, whereas the cream is more useful for vulval and anal lesions.
Imiquimod 5% cream self-application can be used for keratinised lesions and non-keratinised external lesions. It should be applied thinly three times a week at night until the lesions resolve, for a maximum of 16 weeks.
Trichloracetic acid (TCA) is less used due to its corrosive action on skin but is occasionally employed by hospital specialists for warts that are very indurated or in pregnant patients in whom other agents are contra-indicated. This is applied by the specialist rather than for self-application.
An ointment containing a mixture of green tea catechins marketed as Veregen® is used in the USA, Austria and Germany but is not currently licensed in the UK12 .
Keratinised lesions may be better treated with physical ablative methods such as cryotherapy, excision, TCA or electrocautery. Injectable local anaesthetic (eg, 2% lidocaine) should be used before any surgical excision or ablative procedure (other than cryotherapy). Topical anaesthetics (eg, lidocaine cream, EMLA®) can also be used prior to local anaesthetic injection. It can also be used prior to cryotherapy, particularly when treating larger lesions.
Efficacy of treatment
Electrosurgery and scissors excision have clearance rates approaching 100%.
Recurrences occur after all therapies.
All treatments are associated with itching, pain, burning and erosions.
All topical treatments can cause local skin reactions and, where severe, patients must stop treatment and seek advice. Normal surrounding skin may be protected from podophyllotoxin by the use of petroleum jelly. Avoid contact with broken skin and open wounds. Unprotected sexual contact soon after application should be avoided (as it may have an irritant effect on the partner).
Podophyllotoxin treatment is associated with a 45-83% clearance rate and 6-55% recurrence rate at 8-12 weeks.
Imiquimod treatment has a 35-68% clearance rate and 6-26% recurrence rate at 10-24 weeks.
Follow-up is required to assess response and side-effects and to check for new lesions. Continue treatment if response is good but new lesions are developing. Change treatment if it is not tolerated or response to treatment by six weeks (8-12 weeks for imiquimod) is poor.
Specific treatment situations
Cervical warts: should be treated by a gynaecologist and require colposcopy1 .
Intra-anal warts: require proctoscopy and possibly surgical referral.
Pregnancy: avoid podophyllotoxin and imiquimod in pregnancy. Cryotherapy is often used to try to minimise lesions present at delivery but risks (perinatal transmission of genital warts, laryngeal papillomatosis, obstructed labour) to the baby are usually considered small and not an indication for caesarean section.
Genital warts in children
The discovery of external genital warts in children often raises concern about sexual abuse13 . National Institute for Health and Care Excellence (NICE) guidance recommends considering sexual abuse in a child younger than 13 years with anogenital warts unless there is clear evidence of14 :
Mother-to-child transmission during birth (it is not currently known at what age this can be confidently excluded).
Non-sexual transmission from a household member.
Studies suggest sexual transmission as the cause of infection in between 31-58% of children with anogenital warts. One study found an increase in HPV infection in children who had been abused15 . However, it is often very difficult to determine the mode of transmission in children, even with viral typing of the index case and contacts, and the presence of warts or HPV infection without supporting social and clinical evidence cannot be taken as diagnostic of sexual abuse16 . Advice should be sought from experienced child protection colleagues.
There is a lack of controlled trials comparing treatments of anogenital warts in children and adolescents13 .
Prognosis17
Explain to patients that untreated external genital warts may:
Resolve spontaneously - up to one third spontaneously regress within six months.
Remain the same.
Increase in size.
Lifelong subclinical infection may persist. Recurrences are common.
Complications include psychological distress, malignant change within existing warts and an increased risk of certain cancers.
Over 90% of genital warts are caused by HPV types which are low-risk for neoplastic transformation. However, there may be co-infection with oncogenic HPV types. The risk of cervical cancer in situ has been shown to be doubled in women with a history of anogenital warts18 . People with anogenital warts have been shown to have an increased risk of anogenital cancers (anal, vaginal, vulval and cervical), head and neck cancers, non-melanoma skin cancers, smoking-related cancers (bladder, lung, liver) and lymphomas19 .
Anogenital warts prevention
Condoms
Data are conflicting but there is evidence of protection against anogenital warts and condom use is generally advised20 .
Vaccination
There are two HPV vaccines available in the UK: Cervarix® and Gardasil®.
Cervarix® is a bivalent vaccine, meaning it protects against two strains of HPV. Cervarix® protects against HPV16 and HPV18 and so is aimed to reduce (in time) the number of cases of cervical cancer. When the UK first started immunising young women against HPV, this was the vaccine chosen.
Gardasil® is a quadrivalent vaccine, meaning it protects against four strains of HPV. Gardasil® protects against HPV16, HPV18 and HPV6 and HPV11. This means that it also protects against genital warts as well as cervical cancer.
In September 2012 the UK HPV vaccination programme switched to using Gardasil®.
HPV vaccines are highly effective at preventing the infection of susceptible women with the HPV types covered by the vaccine. In clinical trials, both vaccines are over 99% effective at preventing pre-cancerous lesions associated with HPV types 16 or 18 in young women21 . Current studies suggest that protection is maintained for at least ten years. See Further Reading section, below. Based on the immune responses, it is expected that protection will be extended further; long-term follow-up studies are in place22 .
From September 2019 the HPV vaccine has also been offered to year 8 boys23 . This is because the evidence is clear that the HPV vaccine helps protect both boys and girls from HPV-related cancers.
Further reading and references
- Human papillomavirus (HPV): the green book, chapter 18a; UK Health Security Agency, last updated 31 March 2022
- Condyloma Acuminatum; DermIS (Dermatology Information System)
- Dediol I, Buljan M, Vurnek-A Ivkovia M, et al; Psychological burden of anogenital warts. J Eur Acad Dermatol Venereol. 2009 Sep;23(9):1035-8. Epub 2009 Mar 11.
- Genital warts - Images; DermNet NZ
- Lacey CJ, Woodhall SC, Wikstrom A, et al; 2012 European guideline for the management of anogenital warts. J Eur Acad Dermatol Venereol. 2012 Mar 12. doi: 10.1111/j.1468-3083.2012.04493.x.
- Management of anogenital warts; British Association for Sexual Health and HIV (2015)
- Chelimo C, Wouldes TA, Cameron LD, et al; Risk factors for and prevention of human papillomaviruses (HPV), genital warts and cervical cancer. J Infect. 2013 Mar;66(3):207-17. doi: 10.1016/j.jinf.2012.10.024. Epub 2012 Oct 26.
- Jin F, Prestage GP, Kippax SC, et al; Risk factors for genital and anal warts in a prospective cohort of HIV-negative homosexual men: the HIM study. Sex Transm Dis. 2007 Jul;34(7):488-93.
- Wieland U, Kreuter A, Pfister H; Human papillomavirus and immunosuppression. Curr Probl Dermatol. 2014;45:154-65. doi: 10.1159/000357907. Epub 2014 Mar 13.
- Ali H, Donovan B, Wand H, et al; Genital warts in young Australians five years into national human papillomavirus vaccination programme: national surveillance data. BMJ. 2013 Apr 18;346:f2032. doi: 10.1136/bmj.f2032.
- Warts - anogenital; NICE CKS, April 2017 (UK access only)
- Delaney EK, Baguley S; Genital warts. BMJ. 2008 Oct 17;337:a1171. doi: 10.1136/bmj.a1171.
- Korber A, Dissemond J; Pearly penile papules. CMAJ. 2009 Sep 15;181(6-7):397. Epub 2009 Aug 4.
- Wollina U, Verma S; Vulvar vestibular papillomatosis. Indian J Dermatol Venereol Leprol. 2010 May-Jun;76(3):270-2.
- Stanley M; HPV vaccination in boys and men. Hum Vaccin Immunother. 2014;10(7):2109-11. doi: 10.4161/hv.29137.
- Hoy SM; Polyphenon E 10% ointment: in immunocompetent adults with external genital and perianal warts. Am J Clin Dermatol. 2012 Aug 1;13(4):275-81. doi:
- Thornsberry L, English JC 3rd; Evidence-based treatment and prevention of external genital warts in female pediatric and adolescent patients. J Pediatr Adolesc Gynecol. 2012 Apr;25(2):150-4.
- When to suspect child maltreatment; NICE Clinical Guideline (July 2009 - last updated October 2017)
- Unger ER, Fajman NN, Maloney EM, et al; Anogenital human papillomavirus in sexually abused and nonabused children: a multicenter study. Pediatrics. 2011 Sep;128(3):e658-65. Epub 2011 Aug 15.
- Jayasinghe Y, Garland SM; Genital warts in children: what do they mean? Arch Dis Child. 2006 Aug;91(8):696-700. Epub 2006 May 2.
- Leslie SW, Sajjad H, Kumar S; Genital Warts
- Nordenvall C, Chang ET, Adami HO, et al; Cancer risk among patients with condylomata acuminata. Int J Cancer. 2006 Aug 15;119(4):888-93.
- Blomberg M, Friis S, Munk C, et al; Genital warts and risk of cancer: a Danish study of nearly 50 000 patients with genital warts. J Infect Dis. 2012 May 15;205(10):1544-53. doi: 10.1093/infdis/jis228. Epub 2012 Mar 15.
- Lopez-Diez E, Perez S, Carballo M, et al; Lifestyle factors and oncogenic papillomavirus infection in a high-risk male population. PLoS One. 2017 Sep 12;12(9):e0184492. doi: 10.1371/journal.pone.0184492. eCollection 2017.
- Lehtinen M, Dillner J; Clinical trials of human papillomavirus vaccines and beyond. Nat Rev Clin Oncol. 2013 Jul;10(7):400-10. doi: 10.1038/nrclinonc.2013.84. Epub 2013 Jun 4.
- Rana MM, Huhtala H, Apter D, et al; Understanding long-term protection of human papillomavirus vaccination against cervical carcinoma: Cancer registry-based follow-up. Int J Cancer. 2013 Jun 15;132(12):2833-8. doi: 10.1002/ijc.27971. Epub 2012 Dec 19.
- HPV vaccination guide (easy to read leaflet as PDF on HPV vaccination); Public Health England
Article History
The information on this page is written and peer reviewed by qualified clinicians.
Next review due: 26 Oct 2026
27 Oct 2021 | Latest version

Feeling unwell?
Assess your symptoms online for free