Eosinophilic oesophagitis
Peer reviewed by Dr Philippa Vincent, MRCGPLast updated by Dr Colin Tidy, MRCGPLast updated 21 Jul 2023
Meets Patient’s editorial guidelines
- DownloadDownload
- Share
- Language
- Discussion
- Audio Version
Medical Professionals
Professional Reference articles are designed for health professionals to use. They are written by UK doctors and based on research evidence, UK and European Guidelines. You may find the Eosinophilic oesophagitis article more useful, or one of our other health articles.
In this article:
Eosinophilic oesophagitis (EO) - also abbreviated to EoE in American publications - is a disease characterised by oesophageal symptoms and infiltration of eosinophils in the oesophageal epithelium. Originally thought to be a type of gastro-oesophageal reflux disease (GORD), its failure in many (but not all) patients to respond to proton pump inhibitors (PPIs) and the recognition of eosinophilic infiltration on biopsy eventually led to EO being classified as a disease in its own right.
Continue reading below
Eosinophilic oesophagitis epidemiology1
Eosinophilic oesophagitis (EO) is an increasingly common cause of dysphagia in both children and adults, as well as one of the most prevalent oesophageal diseases with a significant impact on physical health and quality of life.
Eosinophilic oesophagitis is more common in men than women and in people of white ethnic origin compared with other ethnic groups.
Having an affected first-degree relative increases the risk of eosinophilic oesophagitis.
The incidence rises during adolescence and peaks in early adulthood.
Eosinophilic oesophagitis symptoms (presentation)1
Back to contentsEosinophilic oesophagitis is characterised by symptoms of dysphagia and/or food impaction in adults, and feeding problems, failure to thrive, abdominal pain and/or vomiting in children.
There is seasonal variation in the symptoms of eosinophilic oesophagitis in many patients, which seems to be associated with higher pollen counts.
OE tends to be a late diagnosis and is often mistaken for GORD in the early stages. It may only be when dysphagia or food bolus obstruction develops that appropriate investigations are instituted and the true diagnosis revealed.
Continue reading below
Associated diseases
Back to contentsA personal history of allergy is common including:
Oral allergy syndrome.
Differential diagnosis2
Back to contentsKnown causes of tissue eosinophilia need to be excluded. These include:
Infections.
Malignancy.
Collagen vascular diseases.
Hypersensitivity.
Inflammatory bowel disease.
Continue reading below
Investigations1 2
Back to contentsOesophageal histology shows a peak eosinophil count of ≥15 eosinophils/high power field (or ≥15 eosinophils/0.3 mm2 or >60 eosinophils/mm2, in the absence of other causes of oesophageal eosinophilia.
Endoscopy and biopsy to exclude eosinophilic oesophagitis should be undertaken in children with typical gastro-oesophageal reflux disease symptoms refractory to treatment with proton pump inhibitors.
Endoscopy and biopsies to exclude eosinophilic oesophagitis in adult patients with typical gastro-oesophageal reflux disease symptoms refractory to proton pump inhibitors is usually not indicated, given the low prevalence of eosinophilic oesophagitis in such patients, in the absence of clinical features associated with eosinophilic oesophagitis (eg, dysphagia or atopy).
For an accurate diagnosis of eosinophilic oesophagitis, proton pump inhibitors should be withdrawn for at least 3 weeks prior to endoscopy and biopsy.
Endoscopic abnormalities of the oesophagus may include:
Diminished vascular pattern.
Mucosal furrows.
Thick mucosa.
Exudates.
Strictures.
Rings - as in image below.
Laryngeal oedema, vocal cord nodules, laryngeal ventricular obliteration.
Multi-ring oesophagus
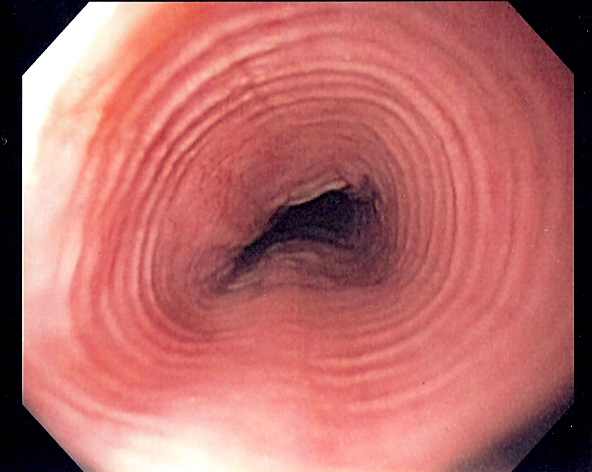
By Samir (own work) via Wikimedia Commons
All adults undergoing endoscopy should have oesophageal biopsies taken if they have endoscopic signs associated with eosinophilic oesophagitis, or symptoms of dysphagia or food bolus obstruction, with a normal looking oesophagus. All children undergoing endoscopy for upper gastrointestinal symptoms should have oesophageal biopsies taken to diagnose eosinophilic oesophagitis.
Histological abnormalities may include:
Abnormally long papillae.
Fibrotic lamina propria.
Microabscesses.
Eosinophilic granules.
Increased extracellular major basic protein (MBP).
Oesophageal physiological testing should be considered in patients with eosinophilic oesophagitis who have ongoing dysphagia, despite histological remission and the absence of fibrostenotic disease at endoscopy.
Other tests may be required to exclude other causes of tissue eosinophilia, depending on the clinical presentation.
Eosinophilic oesophagitis treatment and management1
Back to contentsDiagnosing and treating eosinophilic oesophagitis effectively early in its natural history may prevent long-term complications of fibrosis and strictures requiring subsequent endoscopic intervention.
In patients with food bolus obstruction, urgent referral to gastroenterology and an endoscopy on the next available endoscopy list, or as an immediate emergency is recommended, depending on clinical presentation.
After initiation of therapy (dietary or pharmacological treatment), endoscopy with biopsy while on treatment is recommended to assess response, as symptoms may not always correlate with histological activity.
Dietary treatment:
Exclusive elemental diets have a limited role in eosinophilic oesophagitis, with high efficacy but low compliance rates and should be reserved for patients refractory to other treatments.
Elimination diets are effective in achieving clinico-histological remission in both adults and paediatric patients with eosinophilic oesophagitis.
A six food elimination diet results in higher histological remission rates than two or four food elimination diets, but is associated with lower compliance and an increased number of endoscopies.
When undertaking a dietary restriction therapy for eosinophilic oesophagitis, support from an experienced dietitian throughout both the elimination and reintroduction process is strongly recommended.
Allergy testing to foods (eg, skin prick, specific IgE and patch testing) is not recommended for choosing the type of dietary restriction therapy for eosinophilic oesophagitis.
Combining elimination diets with pharmacological treatment is not routinely recommended but can be considered in cases of drug treatment failure.
Pharmacological treatment:
Proton pump inhibitor therapy is effective in inducing histological and clinical remission in patients with eosinophilic oesophagitis. Proton pump inhibitor therapy should be given two times per day for at least 8–12 weeks prior to assessment of histological response, while on treatment. In patients who achieve histological response, proton pump inhibitor therapy appears effective in maintaining remission.
Topical steroids are effective for inducing histological and clinical remission in eosinophilic oesophagitis. Clinical and histological relapse is high after withdrawal of topical steroid treatment, and following clinical review, maintenance treatment should be recommended. Systemic steroids are not recommended in eosinophilic oesophagitis.
The following are not recommended:
Immunomodulators (eg, azathioprine, 6-mercaptopurine).
Monoclonal antibody therapies, such as anti-tumour necrosis factor (TNF) and anti-integrin therapies.
Sodium cromoglycate, montelukast and antihistamines are not recommended in the management of eosinophilic oesophagitis but may have a role in concomitant atopic disease.
Novel biologics used in other allergic conditions (such as dupilumab, cendakimab and benralizumab) have shown promise in the treatment of eosinophilic oesophagitis.
If symptoms recur while on treatment, endoscopy should be repeated for assessment and to obtain further histology.
Patients with eosinophilic oesophagitis refractory to treatment and/or with significant concomitant atopic disease should be jointly managed by a gastroenterologist and specialist allergist to optimise treatment.
Dietary manipulation
Dietary manipulation is a non-pharmacological treatment which can be effective both in children and in adults. There are three main approaches:
Elemental diet: this involves taking an amino acid mixture for six weeks. The mixture is often considered unpalatable and compliance is often poor but considerable improvement in symptoms and the histological picture has been noted.
Exclusion of six food groups.
Exclusion of food based on allergen tests.
It is essential to involve a dietician and sometimes an allergist if dietary manipulation is attempted.
Topical steroids3
This is the first-line medical treatment in adults and children. Fluticasone metered dose inhaler is usually used. The inhaler is sprayed into the mouth without inhaling and dry swallowed. Patients should be advised not to eat food or drink liquid for half an hour after a dose. Budesonide oral solution is an alternative, mixed with sucralose, chocolate syrup or honey. The treatment is effective in promoting histological remission but a systematic review suggested this does not always result in symptomatological improvement. Only 1% of steroid is absorbed so systemic side-effects are rare. Oral or oesophageal candidiasis are, however, common side-effects.
Orodispersible budesonide tablets
The National Institute for Health and Care Excellence (NICE) has published new Technology Appraisal Guidance,4 recommending budesonide as an orodispersible tablet (ODT) as an option for inducing remission of eosinophilic oesophagitis in adults.
Endoscopic treatment1
Endoscopic dilatation is effective in improving symptoms in patients with fibrostenotic disease due to eosinophilic oesophagitis. Endoscopic dilatation is safe in patients with eosinophilic oesophagitis and can be performed using either balloon or bougie dilators.
Complications5
Back to contentsStricture
Left untreated, eosinophilic oesophagitis is progressively more likely to end up with stricturing disease. About 10% of patients seem to suffer strictures.
Acute food bolus obstruction
This requires immediate attendance at an emergency department. No particular therapy is effective other than arranging an endoscopy to remove the food bolus. This may involve extraction of the bolus or pushing the bolus into the stomach.
Pushing the bolus into the stomach should only be undertaken gently and where there is a lumen available to allow passage of the bolus and the endoscope. There is a risk of perforation with this manoeuvre but this has been rarely reported.
Spontaneous perforation
Eosinophilic oesophagitis is now the commonest disease underlying spontaneous perforation of the oesophagus. Such a perforation is mainly just air and fluid passing through a frayed muscular wall, not food or solid material leaving the lumen of the oesophagus.
The size of the perforation is usually small and the character of the perforation multiple localised points of mural disruption.
Once this has been diagnosed by contrast radiology then the usual therapy is stent, antibiotics and intravenous feeding until the signs of mediastinal inflammation have settled down.
Surgical resection or repair of such perforations has very rarely been reported.
Prognosis6
Back to contentsOE is a chronic disease causing inflammation of the oesophagus. The condition is usually diagnosed several years after onset.
Continued maintenance pharmacological therapy and/or dietary manipulation helps to reduce complications, particularly in patients who have a history of food impaction, dysphagia or oesophageal stricture.
Maintenance is also particularly important in patients with a history of rapid histological and/or symptomatic relapse after stopping initial treatment.
Further reading and references
- Gonsalves NP, Aceves SS; Diagnosis and treatment of eosinophilic esophagitis. J Allergy Clin Immunol. 2020 Jan;145(1):1-7. doi: 10.1016/j.jaci.2019.11.011.
- Dhar A, Haboubi HN, Attwood SE, et al; British Society of Gastroenterology (BSG) and British Society of Paediatric Gastroenterology, Hepatology and Nutrition (BSPGHAN) joint consensus guidelines on the diagnosis and management of eosinophilic oesophagitis in children and adults. Gut. 2022 Aug;71(8):1459-1487. doi: 10.1136/gutjnl-2022-327326. Epub 2022 May 23.
- Gupte AR, Draganov PV; Eosinophilic esophagitis. World J Gastroenterol. 2009 Jan 7;15(1):17-24.
- Murali AR, Gupta A, Attar BM, et al; Topical steroids in eosinophilic esophagitis: Systematic review and meta-analysis of placebo-controlled randomized clinical trials. J Gastroenterol Hepatol. 2016 Jun;31(6):1111-9. doi: 10.1111/jgh.13281.
- Budesonide orodispersible tablet for inducing remission of eosinophilic oesophagitis; NICE Technology appraisal guidance, June 2021
- Attwood SE; Overview of eosinophilic oesophagitis. Br J Hosp Med (Lond). 2019 Mar 2;80(3):132-138. doi: 10.12968/hmed.2019.80.3.132.
- Muir A, Falk GW; Eosinophilic Esophagitis: A Review. JAMA. 2021 Oct 5;326(13):1310-1318. doi: 10.1001/jama.2021.14920.
Continue reading below
Article history
The information on this page is written and peer reviewed by qualified clinicians.
Next review due: 19 Jul 2028
21 Jul 2023 | Latest version

Ask, share, connect.
Browse discussions, ask questions, and share experiences across hundreds of health topics.

Feeling unwell?
Assess your symptoms online for free