Atrial septal defect
Peer reviewed by Dr Hayley Willacy, FRCGP Last updated by Dr Colin Tidy, MRCGPLast updated 21 Apr 2023
Meets Patient’s editorial guidelines
- DownloadDownload
- Share
- Language
- Discussion
- Audio Version
Medical Professionals
Professional Reference articles are designed for health professionals to use. They are written by UK doctors and based on research evidence, UK and European Guidelines. You may find one of our health articles more useful.
In this article:
Continue reading below
What is atrial septal defect?
Atrial septal defect (ASD) is a type of heart defect which allows communication between the left and right side of the heart. These interatrial communications can result from defects in the interatrial septum but also from defects in the atrial connections of the systemic or pulmonary veins (sinus venosus or coronary sinus defects).
Patent foramen ovale is a common congenital atrial septal defect with an incidence of 15-35% in the adult population. The development of the interatrial septum is completed only after birth. In 75% of the general population, the foramen ovale closes after birth, and only an oval depression, called fossa ovalis, remains on the right side of the interatrial septum. Patent foramen ovale can be associated with various clinically important conditions, including migraine and stroke, or decompression illness in divers.1
How common is atrial septal defect? (Epidemiology)2 3
Back to contentsASDs are the third most common congenital heart disease and occur as an isolated anomaly in 5% to 10% of all congenital heart defects. About 30% to 50% of children with congenital heart defects have an ASD as part of their cardiac defect. With improved detection of silent defects by echocardiography the incidence seems to be increasing.
The incidence is higher in females and with the common ostium secundum defects the female to male ratio is 2:1.
Continue reading below
Associated conditions
Back to contentsMost ASDs are sporadic with no defined cause. However, familiar clusters of secundum ASDs have been reported with various forms of inheritance, most commonly autosomal dominant.
The risk of a secundum ASD is increased in families with a history of congenital heart disease, especially when an ASD is present in a sibling.4
Secundum ASDs are common in many syndromes including Holt-Oram syndrome, Noonan's syndrome and Ellis-van Creveld syndrome. Both secundum and primum ASDs are common in Down's syndrome.5
Fetal alcohol syndrome, maternal smoking in the first trimester and maternal diabetes can all be associated with ASDs.
Classification6
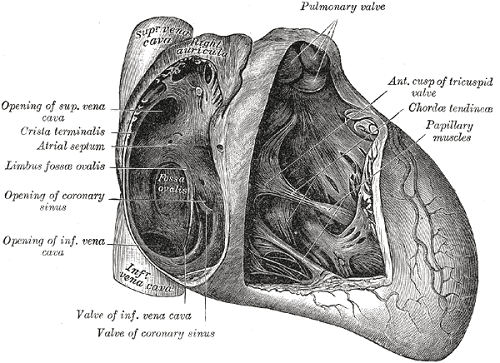
Back to contentsDevelopment of the atrial septum is a complex process and forms the basis of classification of interatrial communications. Atrial septation involves septum primum, septum secundum and atrioventricular canal septum.
Normal development of the atrial septum results in formation of fossa ovalis, an oval shaped depression between the septum secundum and septum primum. Other parts of the septum include sinus venosus septum and coronary sinus septum.
The following types of interatrial communications are described
Patent foramen ovale - this is a normal communication in fetal life and is the space between an appropriately developed septum primum and normal septum secundum.
Secundum ASD - this is the most common type of ASD accounting for 50% to 70% of all ASDs. The secundum ASD is a defect within the fossa ovalis resulting from single or multiple defects in the septum primum.
Primum ASD - this is one of the variants of atrioventricular septal defects (AVSDs) and is also known as partial AVSD as it is associated with the presence of a common atrioventricular orifice. The defect in the atrial septum lies between the inferior margin of fossa ovalis and atrioventricular valves. Isolated primum ASD occurs in 15% of all ASDs.
Sinus venosus defect - the tissue that separates the right pulmonary veins from the superior vena cava and adjacent free atrial wall is called sinus venosus and a defect in this area results in a sinus venosus defect. This accounts for about 10% of all ASDs.
Coronary sinus defect - this results from partial or complete unroofing of the tissue separating coronary sinus from the left atrium, allowing a shunt through the defect and coronary sinus orifice.
interatrial septum
Continue reading below
Atrial septal defect symptoms (presentation)3
Back to contentsMost children with isolated atrial septal defects are free of symptoms, but the rates of exercise intolerance, atrial tachyarrhythmias, right ventricular dysfunction, and pulmonary hypertension increase with advancing age, and life expectancy is reduced in adults with untreated defects.
Infants and children with ASDs are usually asymptomatic.
Rarely, infants may present with tachypnoea, poor weight gain and recurrent chest infections. In all such cases a thorough search should be done for non-cardiac conditions and pulmonary hypertension.7
Subtle symptoms of shortness of breath with exercise or palpitations may occur in some patients in the second decade of life.
Most adult patients with a large defect present with symptoms including fatigue, exercise intolerance, palpitations, syncope, shortness of breath, peripheral oedema, thromboembolic manifestations, and cyanosis.
Arrhythmias are uncommon in children but increasingly common after 40 years of age. The most common arrhythmias are atrial flutter and atrial fibrillation.
Pulmonary hypertension is uncommon in children with isolated ASD but mild or moderate pulmonary hypertension is common in adults with large defects and tends to increase with age.
Pulmonary vascular obstructive disease with or without reversal of atrial shunt (Eisenmenger's syndrome) develops in 5% to 10% of adults with untreated ASDs.8
Physical signs of atrial septal defect9
Back to contentsMost infants and children with an ASD appear well and pink.
Characteristic auscultatory findings are described as:
Widely split second heart sound without respiratory variation.
Soft systolic ejection murmur (Grade 2 to 3) in the pulmonary area at the upper left sternal border.
A diastolic rumble over the left lower sternum because of increased flow through the tricuspid valve in those with a large shunt.
Classic auscultatory findings as described above are not present unless the shunt is reasonably large. The findings may be absent in many infants and children, even in those with a large defect, if the right ventricle is poorly compliant. Classic auscultatory findings are much more likely to be present in adults with large shunts.
Signs of right heart failure are rare but may be seen in adults with pulmonary hypertension.
Investigations9 10
Back to contentsECG
ECG may be normal in infants and children with small shunts
Typical findings include a tall P wave indicating right atrial enlargement, incomplete right bundle branch block pattern (rsR' in V1), and right axis deviation.
Left axis deviation with a superior axis suggests a primum defect.
Right ventricle hypertrophy is seen in patients with pulmonary hypertension.
In adults, atrial flutter or atrial fibrillation may be present.
CXR
CXR is normal in infants and children with small shunts.
In patients with haemodynamically significant ASDs cardiomegaly with enlargement of right atrium and right ventricle may be present.
A prominent pulmonary artery segment and increased pulmonary vascular markings are seen in those with a significant shunt.
Echocardiography
Transthoracic echocardiography is usually diagnostic and is able to show the position, size, and haemodynamic characteristics of the defect. Two-dimensional imaging shows the location and size of the ASD while the Doppler (colour and pulse) provides direction of flow and flow velocity. Measurement of right atrial, right ventricular and pulmonary artery size helps in assessing the haemodynamic impact.
Three-dimensional echocardiography can provide additional information about the defect's shape and change in size during the cardiac cycle.
In older children and adults, especially those who are overweight, adequate visualisation of the atrial septum may not be possible with transthoracic echocardiography. In such cases, transoesophageal echocardiography (TOE) is very useful.
Contrast echocardiography with injection of agitated saline through a peripheral venous line can assist in the diagnosis of ASD in patients with restricted acoustic windows.11
MRI and CT
Cardiac MRI can be used to accurately define the anatomy of ASDs and quantitatively assess their haemodynamic impact. Although it is rarely needed for isolated secundum or primum ASDs, cardiac MRI is a very useful diagnostic tool for sinus venosus ASD which can be difficult to evaluate with echocardiography.12
High-resolution contrast CT can provide excellent anatomical information but is rarely used because of associated radiation risk.13
Cardiac catheterisation
Catheterisation is rarely used as a diagnostic tool for ASDs and is usually done to carry out interventional closure of the defect.
Diagnostic catheterisation is indicated in patients with pulmonary hypertension and in adults at risk of coronary artery disease.
Differential diagnosis
Back to contentsDiagnosis of ASD is often delayed as the murmur can be quite soft and easily mistaken for an Innocent murmur.
Pulmonary stenosis murmur is quite similar.
Atrial septal defect treatment and management14
Back to contentsSurgical closure is safe and effective and when done before age 25 years is associated with normal life expectancy. When asymptomatic, closure is usually performed at 4 to 5 years of age. While there has been some controversy regarding ASD closure in adults, it is recommended that ASD be closed at the time of presentation.
Transcatheter closure offers a less invasive alternative for patients with a secundum defect who fulfil anatomical and size criteria.3 For ostium secundum defects, transcatheter closure has been shown to be as effective as surgical closure. The remaining types of ASDs usually require surgical correction.
Medical management
In children with ASDs, exercise restriction is not needed.
Infective endocarditis prophylaxis is not indicated unless there is associated mitral valve prolapse or other associated defects. Patients with isolated ASD are not considered at risk of developing infective endocarditis.15
Treatment with diuretics may be needed in the small number of children who develop heart failure.
A haemodynamically significant ASD should be closed electively on confirmation of diagnosis. Although this is possible even in small infants, current UK practice is to close the defects at about 4-5 years of age.
Adults with late diagnosis and unrepaired defects may need treatment for heart failure and atrial flutter or atrial fibrillation.
Surgical closure16
ASDs which can only be closed by surgical means include sinus venosus, coronary sinus and primum septal defects. Secundum ASDs can be closed either by surgical means or by transcatheter device. In small infants and in the case of very large defects device closure may not be possible.
Surgical closure is done under direct vision using cardiopulmonary bypass, either by direct suture or by using a pericardial or synthetic patch. Various surgical approaches are possible, including use of a midline sternal incision and lateral thoracotomy.
Video-assisted thoracoscopic procedures are feasible and permit surgery via a much smaller incision, with better cosmetic results.17
Surgical closure of ASDs has been a standard modality of treatment for decades. Since the first reports of surgical ASD closure in 1948, more than 70 years of experience have resulted in a safe and effective surgery with minimal mortality and complications.18
Transcatheter closure19
Transcatheter closure has become an accepted alternative to surgical repair for ostium secundum atrial septal defects. However, large ASDs (>38 mm) and defects with deficient rims are usually referred for surgical closure. Transcatheter closure also remains controversial for other complicated ASDs with comorbidities, additional cardiac features and in small children.20
Since the first transcatheter closure of ASD in 1976 there has been significant advancement in this area with the development of several new devices and delivery systems. In many centres transcatheter closure has replaced surgery as the preferred method.
Not all ASDs can be closed via this method and relative contra-indications include:21
Defects with a maximum diameter of more than 40 mm.
Defects with inadequate margins to support the device.
Interference of the device with atrioventricular valve function or pulmonary or systemic venous drainage.
Recent data confirm that device closure of secundum ASDs is safe and effective in children, with technical success in 96% and occlusion rate of 99.6% at 24 hours.22 Very similar good outcomes have been noted in adults undergoing transcatheter closure of ASDs. The National Institute for Health and Care Excellence (NICE) has issued detailed guidance and supports the use of this procedure.23
Complication rates are low and estimated to be about 1.1% for major complications (device embolisation requiring surgery or pericardial tamponade) and 4.8% for minor complications (atrial arrhythmias, vascular complications and transient heart block).22
Late complications are rare but atrial arrhythmias, stroke, device thrombosis, device erosion through the atrial wall or aortic root, device embolisation and death have all been reported.
Studies comparing transcatheter and surgical closure in terms of outcome, efficacy, complications and costs have shown that both methods are at par.24
Endocarditis prophylaxis15
This is not required in isolated ASDs.
Prognosis25
Back to contentsIsolated atrial septal defects repaired in childhood have excellent prognosis with survival similar to that of the general population.
Isolated ASDs, if not diagnosed until late adulthood, result in increasing symptoms of fatigue, shortness of breath, palpitations and manifestations of heart failure with reduction in survival. The shunt worsens in adults with decreased left ventricular compliance because of coronary heart disease or hypertension. Atrial flutter and atrial fibrillation are also common.
The risk of development of pulmonary vascular disease, a potentially lethal complication, is higher in female patients and in older adults with untreated defects.3
Repair of ASDs improves prognosis, even when it is done after the age of 40 years, with reduction in morbidity and mortality. There is general improvement in symptoms and exercise capacity. However, the risk of atrial flutter and atrial fibrillation remains high.
Atrial septal defect and pregnancy
Back to contentsIn women with isolated ASDs (repaired and unrepaired), pregnancy is not associated with signficant increase in maternal complications. There is a low risk of arrhythmias and transient ischaemic attacks. In women with unrepaired defects, the risk of pre-eclampsia, fetal loss and low birth weight is higher than that of the general population.26
Maternal mortality is remarkably high in women with an ASD and severe pulmonary hypertension. Pregnancy should be avoided in such individuals.27
Further reading and references
- Le Gloan L, Legendre A, Iserin L, et al; Pathophysiology and natural history of atrial septal defect. J Thorac Dis. 2018 Sep;10(Suppl 24):S2854-S2863. doi: 10.21037/jtd.2018.02.80.
- Romano V, Gallinoro CM, Mottola R, et al; Patent Foramen Ovale-A Not So Innocuous Septal Atrial Defect in Adults. J Cardiovasc Dev Dis. 2021 May 25;8(6):60. doi: 10.3390/jcdd8060060.
- Hoffman JI, Kaplan S; The incidence of congenital heart disease. J Am Coll Cardiol. 2002 Jun 19;39(12):1890-900.
- Geva T, Martins JD, Wald RM; Atrial septal defects. Lancet. 2014 May 31;383(9932):1921-32. doi: 10.1016/S0140-6736(13)62145-5. Epub 2014 Apr 8.
- Caputo S, Capozzi G, Russo MG, et al; Familial recurrence of congenital heart disease in patients with ostium secundum atrial septal defect. Eur Heart J. 2005 Oct;26(20):2179-84. Epub 2005 Jun 24.
- Dennis J, Archer N, Ellis J, et al; Recognising heart disease in children with Down syndrome. Arch Dis Child Educ Pract Ed. 2010 Aug;95(4):98-104. doi: 10.1136/adc.2007.126672.
- Briggs LE, Kakarla J, Wessels A; The pathogenesis of atrial and atrioventricular septal defects with special emphasis on the role of the dorsal mesenchymal protrusion. Differentiation. 2012 Jul;84(1):117-30. doi: 10.1016/j.diff.2012.05.006. Epub 2012 Jun 17.
- Goetschmann S, Dibernardo S, Steinmann H, et al; Frequency of severe pulmonary hypertension complicating "isolated" atrial septal defect in infancy. Am J Cardiol. 2008 Aug 1;102(3):340-2. doi: 10.1016/j.amjcard.2008.03.061. Epub 2008 May 2.
- Sachweh JS, Daebritz SH, Hermanns B, et al; Hypertensive pulmonary vascular disease in adults with secundum or sinus venosus atrial septal defect. Ann Thorac Surg. 2006 Jan;81(1):207-13.
- Martin SS, Shapiro EP, Mukherjee M; Atrial septal defects - clinical manifestations, echo assessment, and intervention. Clin Med Insights Cardiol. 2015 Mar 23;8(Suppl 1):93-8. doi: 10.4137/CMC.S15715. eCollection 2014.
- Torres AJ; Hemodynamic assessment of atrial septal defects. J Thorac Dis. 2018 Sep;10(Suppl 24):S2882-S2889. doi: 10.21037/jtd.2018.02.17.
- Soliman OI, Geleijnse ML, Meijboom FJ, et al; The use of contrast echocardiography for the detection of cardiac shunts. Eur J Echocardiogr. 2007 Jun;8(3):S2-12. Epub 2007 Apr 25.
- Prompona M, Muehling O, Naebauer M, et al; MRI for detection of anomalous pulmonary venous drainage in patients with sinus venosus atrial septal defects. Int J Cardiovasc Imaging. 2011 Mar;27(3):403-12. doi: 10.1007/s10554-010-9675-3. Epub 2010 Aug 5.
- Johri AM, Rojas CA, El-Sherief A, et al; Imaging of atrial septal defects: echocardiography and CT correlation. Heart. 2011 Sep;97(17):1441-53. doi: 10.1136/hrt.2010.205732.
- Rao PS, Harris AD; Recent advances in managing septal defects: atrial septal defects. F1000Res. 2017 Nov 22;6:2042. doi: 10.12688/f1000research.11844.1. eCollection 2017.
- Prophylaxis against infective endocarditis: Antimicrobial prophylaxis against infective endocarditis in adults and children undergoing interventional procedures; NICE Clinical Guideline (March 2008 - last updated July 2016)
- Liava'a M, Kalfa D; Surgical closure of atrial septal defects. J Thorac Dis. 2018 Sep;10(Suppl 24):S2931-S2939. doi: 10.21037/jtd.2018.07.116.
- Suematsu Y, Kiaii B, Bainbridge DT, et al; Robotic-assisted closure of atrial septal defect under real-time three-dimensional echo guide: in vitro study. Eur J Cardiothorac Surg. 2007 Oct;32(4):573-6. Epub 2007 Aug 15.
- Noiri JI, Konishi H, Matsuzoe H; The importance of pre-ablation atrial septal evaluation for a patient with surgical patch closure history. J Arrhythm. 2023 Feb 20;39(2):224-226. doi: 10.1002/joa3.12829. eCollection 2023 Apr.
- O'Byrne ML, Levi DS; State-of-the-Art Atrial Septal Defect Closure Devices for Congenital Heart. Interv Cardiol Clin. 2019 Jan;8(1):11-21. doi: 10.1016/j.iccl.2018.08.008.
- Fraisse A, Latchman M, Sharma SR, et al; Atrial septal defect closure: indications and contra-indications. J Thorac Dis. 2018 Sep;10(Suppl 24):S2874-S2881. doi: 10.21037/jtd.2018.08.111.
- Marie Valente A, Rhodes JF; Current indications and contraindications for transcatheter atrial septal defect and patent foramen ovale device closure. Am Heart J. 2007 Apr;153(4 Suppl):81-4.
- Everett AD, Jennings J, Sibinga E, et al; Community use of the amplatzer atrial septal defect occluder: results of the multicenter MAGIC atrial septal defect study. Pediatr Cardiol. 2009 Apr;30(3):240-7. doi: 10.1007/s00246-008-9325-x. Epub 2008 Nov 18.
- Endovascular closure of atrial septal defect; NICE Interventional procedures guidance, October 2004
- Kutty S, Hazeem AA, Brown K, et al; Long-term (5- to 20-year) outcomes after transcatheter or surgical treatment of hemodynamically significant isolated secundum atrial septal defect. Am J Cardiol. 2012 May 1;109(9):1348-52. doi: 10.1016/j.amjcard.2011.12.031. Epub 2012 Feb 13.
- Lindsey JB, Hillis LD; Clinical update: atrial septal defect in adults. Lancet. 2007 Apr 14;369(9569):1244-6.
- Drenthen W, Pieper PG, Roos-Hesselink JW, et al; Outcome of pregnancy in women with congenital heart disease: a literature review. J Am Coll Cardiol. 2007 Jun 19;49(24):2303-11. Epub 2007 Jun 4.
- Bowater SE, Thorne SA; Management of pregnancy in women with acquired and congenital heart disease. Postgrad Med J. 2010 Feb;86(1012):100-5.
Continue reading below
Article history
The information on this page is written and peer reviewed by qualified clinicians.
Next review due: 19 Apr 2028
21 Apr 2023 | Latest version
26 Jan 2015 | Originally published
Authored by:
Dr Anjum Gandhi, FRCPCH

Ask, share, connect.
Browse discussions, ask questions, and share experiences across hundreds of health topics.

Feeling unwell?
Assess your symptoms online for free