Lumbar puncture
Peer reviewed by Dr Laurence KnottLast updated by Dr Colin Tidy, MRCGPLast updated 15 Dec 2021
Meets Patient’s editorial guidelines
- DownloadDownload
- Share
- Language
- Discussion
- Audio Version
Medical Professionals
Professional Reference articles are designed for health professionals to use. They are written by UK doctors and based on research evidence, UK and European Guidelines. You may find the Lumbar puncture article more useful, or one of our other health articles.
In this article:
Diagnostic lumbar puncture is one of the most commonly performed invasive tests in hospital clinical medicine. Evaluation of an acute headache and investigation of inflammatory or infectious disease of the nervous system are the most common indications. Serious complications are rare, and correct technique will minimise diagnostic error and maximise patient comfort1 .
Continue reading below
When to do a lumbar puncture
Lumbar puncture (LP) can confirm or exclude meningitis.
Cerebrospinal fluid (CSF) culture is the gold standard in the diagnosis of bacterial meningitis.
More than 90% of cases of acute bacterial meningitis present with a CSF white cell count of more than 100 cells per μL.
In immunocompromised patients, CSF white cell counts are often low.
Polymorphonuclear cells may predominate in cases of non-meningococcal meningitis
Acellular CSF is rare, except in patients with tuberculous meningitis.
Initial Gram staining shows organism in 69-93% of cases of pneumococcal meningitis, and 30-89% of cases of meningococcal meningitis in adults and children.
It also allows identification of uncommon pathogens and viruses by polymerase chain reaction (PCR).
Suspected intracranial bleeding and subarachnoid haemorrhage (SAH):3
LP must be performed in a case of clinically suspected intracranial bleed or SAH if CT or MRI does not confirm the diagnosis.
Within the first 6-12 hours the differentiation between genuine subarachnoid blood and traumatic admixture of blood may be difficult.
To establish diagnosis:
In cases such as confusional states, meningeal malignancies, demyelinating disorders, CNS vasculitis, multiple sclerosis.
To administer medications via the CSF - intrathecal therapy:
Chemotherapy. National guidelines on safe administration of intrathecal chemotherapy were archived in 2013, pending updated guidance. This will reflect changes in chemotherapy and include intrathecal chemotherapy and acute oncology services. Local guidance should be consulted.
Cancer and non-cancer pain4 .
Neuromodulation in spasticity and dystonias5 .
To treat:
Communicating hydrocephalus (also known as normal pressure hydrocephalus).
Benign intracranial hypertension6 .
When not to do a lumbar puncture
Back to contentsContra-indications to LP7
These include:
Signs suggesting raised intracranial pressure:
A reduced or fluctuating level of consciousness (Glasgow Coma Scale score less than 9 or a drop of 3 or more).
Age-relative bradycardia and hypertension.
Focal neurological signs.
Abnormal posture or posturing.
Unequal, dilated or poorly responsive pupils.
Papilloedema.
Abnormal 'doll's eye' movements.
A tense, bulging fontanelle.
Shock.
Extensive or spreading purpura.
Convulsions until stabilised.
Coagulation abnormalities:
Coagulation results (if obtained) outside the normal range.
A platelet count below 100 x 109/litre.
Receiving anticoagulant therapy.
Superficial infection at the LP site.
Respiratory insufficiency:
LP is considered to have a high risk of precipitating respiratory failure in the presence of respiratory insufficiency.
Continue reading below
How to perform a lumbar puncture
Back to contentsBefore the procedure
Explain to the patient what you want to do and why you need to do it. Obtain explicit consent and use the appropriate consent form. Tell the patient that co-operation is important and that they can talk to you at any time.
Explain that you will numb the area with local anaesthetic, which will involve a sharp scratch and then some stinging which will quickly settle. With children aged over 6 months, if there is time, a topical local anaesthetic cream can be used before infiltration.
Advise that, subsequently, there should be no pain. However, they may feel sensations down a lower limb and there could be a pulling/pressure sensation and mild discomfort.
Discuss possible complications (see 'Complications of lumbar puncture', below), give the patient an opportunity to ask questions and remember to document this in the clinical record when you make your notes.
Procedure
Preparing your equipment and the patient properly will save time in the long run and ensure full co-operation.
Place the patient on their left side with their back exactly vertical, aligned with the edge of the bed, with their spine fully flexed - knees up to chin. If you are left-hand dominant, the right lateral position may be easier.
Procedure method
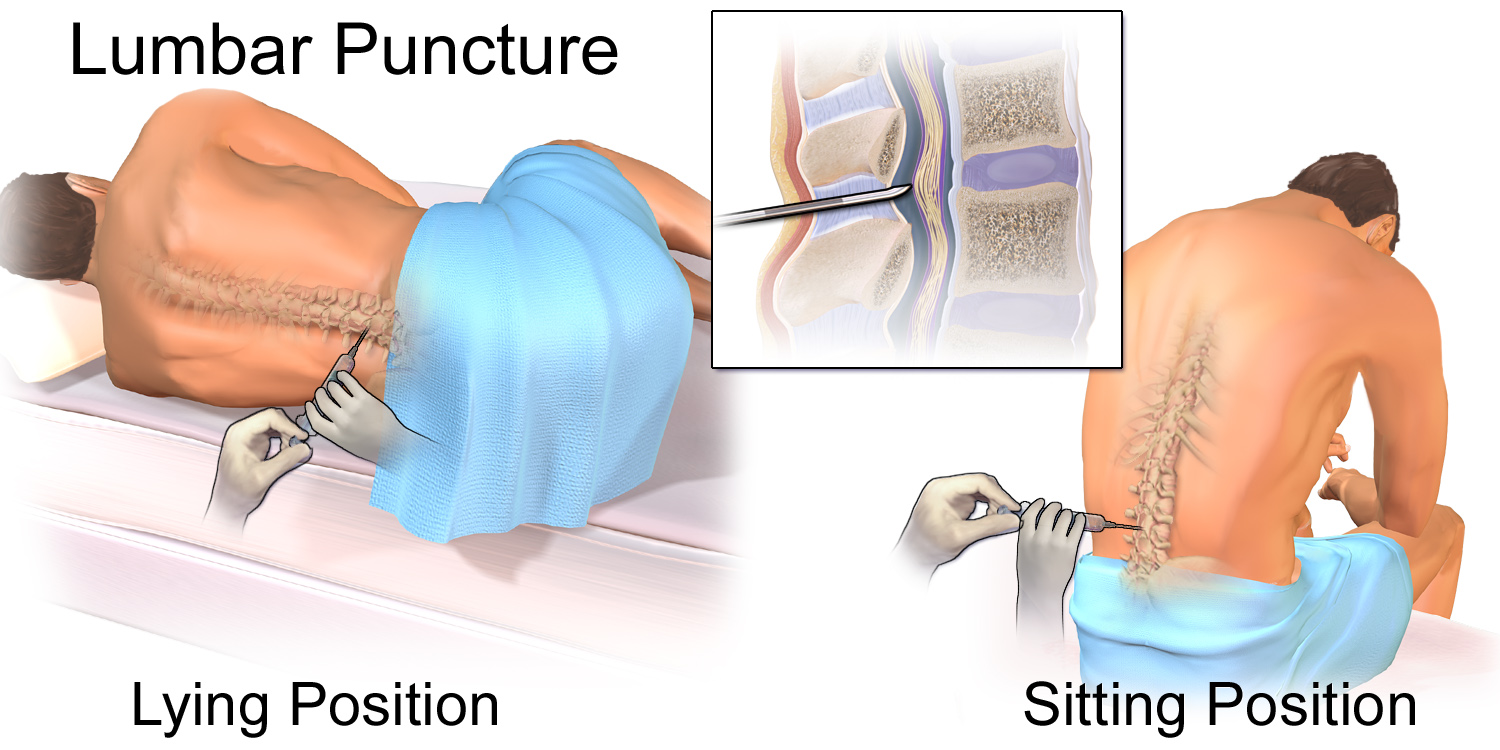
© Blausen.com staff, "Blausen gallery 2014", Wikiversity Journal of Medicine, via Wikimedia Commons
A very useful technique in competent adults is to position the patient so that they are sitting up leaning over a table or pillow, as the anatomy is less distorted. The procedure may be radiologically guided, if necessary.
Mark the intervertebral space L4/L5 or L3/L4 (at the same height as the iliac crest) with a gentle skin indentation using a thumbnail or an object like a pen top.
Wash your hands thoroughly and put on a mask and sterile gloves. From this point on you must observe strict aseptic technique. From the pack, place sterile towels beneath the patient and over their side, to isolate your sterile field.
Open your sterile LP pack and make sure your needle, sample bottles (preferably labelled '1, 2, 3' in the order you will use to collect samples) and manometer are set up and immediately to hand.
Spinal needles are available in varying sizes for different ages - from 30 mm-90 mm usually. Extra-long needles are occasionally required for very large adults.
Sterilise the area with iodine-based antiseptic unless the patient is allergic. If you are using a fenestrated sterile adhesive drape, the sterile field can be isolated at this point.
Anaesthetise the skin with 1% lidocaine. Use a small-gauge needle to minimise pain. Let a minute pass, then infiltrate lidocaine into the interspinous area; in bigger patients use a longer, larger-gauge needle if required. ALWAYS aspirate before injecting lidocaine, to avoid intravascular injection.
After one minute, insert a 22G spinal needle with stylet in place, horizontally through the mark, aiming towards the umbilicus with the needle bevel facing upwards.
Make sure that you are in the midline between the iliac crests and in the interspinous space. Feel resistance from the spinal ligaments and then the dura, and feel 'give' as the needle enters the subarachnoid space.
Withdraw the stylet and wait for CSF to appear at the needle cuff.
Measure CSF pressure with the manometer. Connect tubing from the manometer to the needle cuff and hold the gauge upright. When the meniscus settles, gently tap the tube; if there is no movement then read the pressure in cm H2O from the scale. Pressure is normally 5-20 cm H2O. In general, the CSF pressure increases with body mass index (BMI) and decreases with age after the sixth decade8 9 .
Collect the fluid in five numbered bottles (<5-10 ml in total). Usually 5-10 drops for each sample are sufficient. Three bottles go to microbiology, one to virology, one to biochemistry.
Reinsert the stylet to halt CSF flow. There is evidence that leaving the stylet in situ when removing the needle may reduce the incidence of headache due to dural disruption10 .
Clean, then dress, the site.
After the lumbar puncture procedure
Back to contentsPrescribe simple analgesia as required in case of headache.
There is no evidence that prolonged bed rest after LP has any effect on reducing the incidence of headache11 .
Document the procedure, taking care to note that you explained possible complications to the patient, used aseptic technique and what gauge/type of needle was used. It is also useful to document how easy or difficult the procedure was (particularly if repeat LP is needed) and whether it was a bloody tap, as this can affect the results.
Do not perform repeat LP in neonates who are receiving the antibiotic treatment appropriate to the causative organism and are making a good clinical recovery and/or before stopping antibiotic therapy if they are clinically well7 .
Continue reading below
Complications of lumbar puncture
Back to contentsLP is a relatively safe procedure but some major and minor complications may occur. These include1 :
Post-LP headache.
Infection.
Bleeding.
Cerebral herniation (rare but potentially fatal).
Others (for example, epidermoid tumour, abducens palsy, radicular symptoms and low back pain).
The incidence of these complications may be minimised by avoiding the contra-indictions and following the procedures described above.
Post-LP headache (PLPH)
Headache is the most common complication of LP. It lasts for 2-8 days and occurs in around 40% of patients. It is caused by low CSF pressure due to fluid leakage through the hole (so-called 'dural tap'). Typically, it presents in the upright position and is quickly relieved by lying down. The headache is aggravated by coughing or straining and can be associated with nausea, vomiting, vertigo, tinnitus, hearing loss and diplopia. Neck and low backache are also common.
The diagnosis is essentially clinical, based on the history of an LP, the postural nature and the associated symptoms12 .
Prevention of PLPH
It may be prevented by using a thin LP needle (22 G) with an atraumatic tip.
Epidural saline or morphine, and prophylactic blood patch have been studied; however, there is not consensus on the best preventative practice11 .
There is no evidence that caffeine reduces the incidence of PLPH11 .
Treatment of PLPH
With pain relief and oral fluids.
A Cochrane review has shown caffeine to be helpful13 .
Intravenous fluids may be used (there is no convincing evidence to support their use but they may help when headache is causing nausea).
If PLPH still persists, use an extradural blood patch. This is where 10-20 ml of the patient's blood is injected into the extradural space14 .
Further reading and references
- Lumbar puncture: Technique, indications, contraindications, and complications in adults; UpToDate®
- Hudgins PA, Fountain AJ, Chapman PR, et al; Difficult Lumbar Puncture: Pitfalls and Tips from the Trenches. AJNR Am J Neuroradiol. 2017 Jul;38(7):1276-1283. doi: 10.3174/ajnr.A5128. Epub 2017 Mar 16.
- Doherty CM, Forbes RB; Diagnostic Lumbar Puncture. Ulster Med J. 2014 May;83(2):93-102.
- Brouwer MC, Thwaites GE, Tunkel AR, et al; Dilemmas in the diagnosis of acute community-acquired bacterial meningitis. Lancet. 2012 Nov 10;380(9854):1684-92. doi: 10.1016/S0140-6736(12)61185-4.
- Steiner T, Juvela S, Unterberg A, et al; European Stroke Organization Guidelines for the Management of Intracranial Aneurysms and Subarachnoid Haemorrhage. Cerebrovasc Dis. 2013 Feb 7;35(2):93-112.
- Intrathecal drug delivery for the management of pain and spasticity in adults; British Pain Society (2008)
- Spasticity in children and young people; NICE Clinical Guideline (July 2012, updated Nov 2016)
- Lumbar subcutaneous shunt; NICE Interventional Procedure Guidance, June 2004
- Bacterial meningitis and meningococcal septicaemia: Management of bacterial meningitis and meningococcal septicaemia in children and young people younger than 16 years in primary and secondary care; NICE Clinical Guideline (last updated February 2015)
- Fleischman D, Berdahl JP, Zaydlarova J, et al; Cerebrospinal fluid pressure decreases with older age. PLoS One. 2012;7(12):e52664. doi: 10.1371/journal.pone.0052664. Epub 2012 Dec 26.
- Seehusen DA, Reeves MM, Fomin DA; Cerebrospinal fluid analysis. Am Fam Physician. 2003 Sep 15;68(6):1103-8.
- Deibel M, Jones J, Brown M; Best evidence topic report: reinsertion of the stylet before needle removal in diagnostic lumbar puncture. Emerg Med J. 2005 Jan;22(1):46.
- Kim SR, Chae HS, Yoon MJ, et al; No effect of recumbency duration on the occurrence of post-lumbar puncture headache with a 22G cutting needle. BMC Neurol. 2012 Jan 30;12:1. doi: 10.1186/1471-2377-12-1.
- Ahmed SV, Jayawarna C, Jude E; Post lumbar puncture headache: diagnosis and management. Postgrad Med J. 2006 Nov;82(973):713-6.
- Basurto Ona X, Osorio D, Bonfill Cosp X; Drug therapy for treating post-dural puncture headache. Cochrane Database Syst Rev. 2015 Jul 15;7:CD007887. doi: 10.1002/14651858.CD007887.pub3.
- Lavi R, Rowe JM, Avivi I; Lumbar puncture: it is time to change the needle. Eur Neurol. 2010;64(2):108-13. doi: 10.1159/000316774. Epub 2010 Jul 14.
Continue reading below
Article history
The information on this page is written and peer reviewed by qualified clinicians.
Next review due: 14 Dec 2026
15 Dec 2021 | Latest version

Ask, share, connect.
Browse discussions, ask questions, and share experiences across hundreds of health topics.

Feeling unwell?
Assess your symptoms online for free