Keratoplasty
Peer reviewed by Dr Krishna Vakharia, MRCGPLast updated by Dr Colin Tidy, MRCGPLast updated 22 Sept 2023
Meets Patient’s editorial guidelines
- DownloadDownload
- Share
- Language
- Discussion
- Audio Version
Medical Professionals
Professional Reference articles are designed for health professionals to use. They are written by UK doctors and based on research evidence, UK and European Guidelines. You may find one of our health articles more useful.
In this article:
Synonyms: corneal transplant, corneal graft
Continue reading below
What is a keratoplasty?1 2
Keratoplasty is the procedure whereby abnormal corneal tissue is replaced by a healthy donor cornea.
Corneal blindness is one of the major causes of reversible blindness, which can be managed with transplantation of a healthy donor cornea. It is the most successful organ transplantation in the human body as cornea is devoid of vasculature, minimising the risk of graft rejection.
Corneal transplantation or keratoplasty is the most commonly performed and also the most successful allogenic transplant worldwide.
Some of the major complications associated with a standard corneal transplantation surgery (full-thickness or penetrating keratoplasty), include graft rejection, post-penetrating keratoplasty astigmatism, graft failure, suture-related problems, and long-term side-effects of topical corticosteroids.
In an effort to improve the outcomes and further reduce the complications, especially graft rejection, it was found that all the layers of the cornea don't need to be transplanted in all cases. Recently, techniques of transplanting only the diseased component of the cornea have been developed.
These techniques of selective transplantation of layers of the cornea reduce the amount of allogenic tissue being transplanted, so reducing the risk of rejection. In addition, a single donor tissue can be used for multiple recipients as different layers can be transplanted.
Procedures such as epithelial transplant, Bowman’s layer transplant, anterior lamellar keratoplasty, and posterior lamellar keratoplasty have been developed to replace diseased limbal stem cells, anterior stroma, and endothelium.
Although component keratoplasty is known to reduce the risk of immunological rejection, there are cases that still present with rejection.
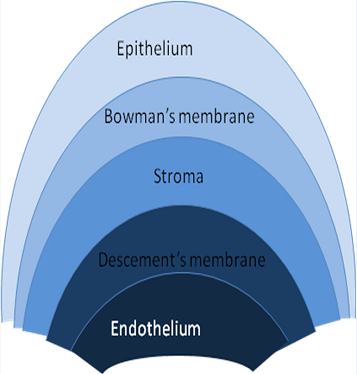
Structure of the cornea2
Back to contentsThe cornea is a multilayered structure consisting of (from outside to inside):
Anterior corneal epithelium:
A thin, multicellular epithelial tissue layer composed of around six layers of cells (non-keratinised stratified squamous epithelium) of rapidly regenerating cells kept moist by tears.
The air/tearfilm interface is the most significant component of the total refractive power of the eye, so disruption of this surface can reduce acuity.
The corneal epithelium is continuous with the conjunctival epithelium. It regenerates constantly, from the bottom layer.
Bowman's layer (also called the anterior limiting membrane):
This is a protective acellular collagen layer.
Corneal stroma (or substantia propria):
A thicker, transparent layer of collagen and keratocytes which makes up around 90% of corneal thickness.
Descemet's membrane (posterior limiting membrane):
A thin acellular layer which acts as the basement membrane to the corneal endothelium and consists mainly of collagen.
There may be an even thinner protective membrane called Dua's layer on its surface. This membrane was described in 2013 but its existence is a matter of dispute.3
Corneal endothelium:
A simple squamous monolayer of cells which regulate fluid and solute transport. These cells do not regenerate but instead stretch to compensate for dead cells.
An ABCDE mnemonic is:
Anterior corneal epithelium, Bowman's membrane, Corneal stroma, Descemet's membrane, Endothelium.
Corneal structure
Continue reading below
Indications2
Back to contentsCorneal blindness is a major cause of global blindness, second only to cataract.4
Indications for penetrating keratoplasty (replacement of the entire cornea) include:
Optical indications - improving acuity by replacing opaque or distorted host tissue with clear tissue. The most common indications are:
Pseudophakic bullous keratopathy.
Keratoglobus.
Corneal dystrophy (eg, Fuchs' endothelial dystrophy): these form the bulk of the older patient group).
Scarring due to keratitis or trauma.
Tectonic indications - to preserve corneal integrity in cases of corneal stromal thinning, corneal perforation or descemetoceles (ulcers extending into the stroma and exposing Descemet's membrane).
Therapeutic indications - this is rare, to remove inflamed corneal tissue which is not responding to other treatments.
Cosmetic indications - where the cornea has a whitish or opaque hue due to scarring.
Lamellar keratoplasty is indicated for:
Opacification of only up to a third of the thickness of the stroma.
Diseases of the corneal margin, such as corneal thinning, recurrent pterygium or limbal dermoids.
Deep lamellar keratoplasty is indicated for disease of the anterior cornea (up to 95% involvement) with sparing of the endothelium, such as in chronic inflammatory disease (which carries a high risk of graft rejection: see 'Complications', below).
The procedures2
Back to contentsKeratoplasty is generally done as a day case. It may be carried out under local or general anaesthetic and may involve an overnight stay. It takes an hour or two to complete and a pad will be placed on the eye until first postoperative review. It is unusual for patients to experience significant pain postoperatively (some swelling and slight discomfort can be expected).
Donor tissue
Donor tissue is harvested within 24 hours of donor death. As with all transplants, there are certain restrictions imposed in order to maximise positive outcome; for example, in common with other transplant procedures, tissue is not accepted where there has been death of unknown cause. In keratoplasty, donors cannot be very young (the cornea is floppy and may give poor refractive results), over 70 years old (there are low endothelial cell counts) or with intrinsic eye disease or previous intraocular surgery.
Penetrating keratoplasty
This begins with determination of the graft size (pre-operatively) and preparation of the donor material. Steps are then:
Excision of diseased host tissue, using a trephine (the rest of the globe contents needs to be protected).
Fixation of donor tissue with interrupted or continuous non-absorbable sutures (to allow adequate tissue healing time).
Finishing: the eye is filled with viscoelastic fluid.
Typically the patient is given antibiotic eye drops and a patch and is discharged the next day.
Lamellar keratoplasty
This is a similar technique but only part of the thickness of the cornea is grafted. This method offers improved tectonic (structural) integrity, although the procedure is technically challenging. The outcome in terms of optical performance may be less good than for full-thickness replacement.5
Deep anterior lamellar keratoplasty
This involves removal of a greater proportion of the anterior corneal layers (leaving the endothelium and Descemet's membrane behind) with a more complicated sequence of corneal layer dissection, trephination and graft placement. This technique is used in cases of anterior corneal opacifications, scars and ectatic diseases such as keratoconus. The procedure is longer than penetrating keratoplasty but is associated with a reduced risk of rejection.
Deep lamellar endothelial keratoplasty (DLEK)
DLEK is a variation in which only the endothelium is replaced.
Descemet stripping endothelial keratoplasty (DSEK)
DSEK has gained widespread acceptance. It combines a simplified technique for stripping off the dysfunctional epithelium from the host cornea with microdissection of the donor tissue. Endothelial keratoplasty replaces the patient's endothelium with a transplanted disc of posterior stroma/Descemet's membrane/endothelium. This reduces ocular surface complications generally compared to penetrating keratoplasty.
Descemet's membrane endothelial keratoplasty (DMEK)
DMEK is a further variation on DSEK, in which the patient's endothelium is replaced with a transplanted disc of Descemet's membrane/endothelium (DMEK). This is a technically more challenging procedure but it reduces immune-mediated rejection by transplanting bare endothelium and Descemet's membrane without stroma. It alleviates the need for long-term topical steroids as an anti-rejection measure and therefore also reduces the incidence of secondary glaucoma.5 6
Note: DESK/DMEK have revolutionised treatment of disorders of the corneal endothelium. Surgery can be performed with only one or two sutures (unlike penetrating keratoplasty). Patients may recover functional vision in days to weeks, as opposed to a year in the case of penetrating (full-thickness) keratoplasty.
Postoperative medical care
Patients are given preservative-free topical steroid/antibiotic drops which are gradually tapered but which may be continued at low doses for a year or more. They may also be given a course of mydriatics. If there was pre-existing herpes simplex keratitis, there may also be an oral course of aciclovir. Oral acetazolamide may be given to those with co-existent glaucoma.
Follow-up is frequent, typically at days 1, 7 and 28, and 2- to 3-monthly thereafter. Removal of sutures is progressive if there are interrupted sutures and is usually completed by about 12 months post-procedure. Wearing of contact lenses may subsequently be required for some patients.
Postoperative non-medical care
Once home, normal bathing and showering can resume but care must be taken not to get water in the eye for a month. If the eye becomes sticky, gentle washing with cooled, boiled water is recommended. Eyelid make-up should also be avoided for this time. Sunglasses can minimise discomfort but contact lenses should be avoided for at least eight weeks. It is very important that the patient does not rub the eye in the early weeks postoperatively. An eye shield will be given to the patient to wear for several weeks whenever they sleep, to avoid inadvertent rubbing.
Swimming should be avoided for at least a month. Contact sports should be avoided until approved by the surgical team. Light work can be resumed in 2-3 weeks and manual labour in 3-4 months. Driving may be affected by a change of visual acuity and the patient should take the team's advice before starting up again. It may take months or even longer to recover useful vision.
Continue reading below
Complications of a keratoplasty1 2
Back to contentsIf any of the complications listed below are suspected, same-day referral is mandatory.
Persistent epithelial defects (>2 weeks in duration): symptoms and signs are as for a corneal abrasion.
Irritation by protruding sutures: presents with a red eye with associated foreign body sensation. Do not evert the eyelids when examining the eye.
Wound leak: look for a shallow anterior chamber and perform a Seidel's test. See separate Examination of the Eye article.
Iris prolapse (through the operative wound): a pigmented mass within the operative wound with an associated pupil deformity.
Uveitis: red eye, photophobia, pain, poor vision ± headaches.
Elevated intraocular pressures: see separate Angle-closure Glaucoma article.
Keratitis or endophthalmitis: rare but the latter is sight-threatening and therefore an emergency.
Late:
Astigmatism (± need for contact lenses or refractive surgery).
Recurrence of the original disease process - this is common with viral keratitis.
Late wound separation and suture-related problems.
Glaucoma.
Cystoid macular oedema.
Added complications of endothelial keratoplasty:
Displacement of the donor tissue requiring repositioning ('refloating'). This is more common with DMEK than with DSEK. Folds in the donor tissue may also reduce the quality of vision, requiring repair.
Gradual reduction in endothelial cell density over time can lead to loss of clarity and require repetition of the procedure.
Early graft rejection
This usually occurs by the first operative day.
There is a cloudy cornea in an otherwise 'quiet' eye (not red, painful, etc).
This is usually due to defective donor endothelium or operative trauma.
Late graft rejection
About 50% occur within the first six postoperative months and the vast majority with a year.
Different types (endothelial versus epithelial) can produce different clinical pictures.
Look for evidence of a red eye, corneal clouding ± uveitis, associated with decreased visual acuity.
This is usually due to immunological graft rejection.
Treatment is with intensive topical steroids ± periocular steroids ± systemic immunosuppression.
Prognosis2
Back to contentsPoor prognosis is noted in patients with:
Additional corneal problems such as vascularisation or peripheral thinning.
Associated ocular disease such as herpes, active inflammation or uncontrolled glaucoma.
Patients with endothelial transplants frequently achieve an average best corrected vision of 20/40 range, although some reach 20/20. Optical irregularity at the graft/host interface may limit vision below 20/20.
Keratoprostheses4
Back to contentsThese are artificial corneal implants. For many patients, corneal transplantation offers a second chance of sight. However, in some cases (eg, multiple graft failure, severe chemical burns and autoimmune diseases such as Stevens-Johnson syndrome and ocular mucous membrane pemphigoid) the ocular environment is too hostile for a corneal graft to survive. Keratoprostheses can offer these patients the prospect of visual rehabilitation.
Dr Mary Lowth is an author or the original author of this leaflet.
Further reading and references
- Armitage WJ, Goodchild C, Griffin MD, et al; High-risk Corneal Transplantation: Recent Developments and Future Possibilities. Transplantation. 2019 Dec;103(12):2468-2478. doi: 10.1097/TP.0000000000002938.
- Deshmukh R, Murthy SI, Rapuano CJ, et al; Graft rejection in component keratoplasty. Indian J Ophthalmol. 2023 Mar;71(3):698-706. doi: 10.4103/ijo.IJO_1964_22.
- Singh R, Gupta N, Vanathi M, et al; Corneal transplantation in the modern era. Indian J Med Res. 2019 Jul;150(1):7-22. doi: 10.4103/ijmr.IJMR_141_19.
- Dua HS, Faraj LA, Said DG, et al; Human corneal anatomy redefined: a novel pre-Descemet's layer (Dua's layer). Ophthalmology. 2013 Sep;120(9):1778-85. doi: 10.1016/j.ophtha.2013.01.018. Epub 2013 May 25.
- Shalaby Bardan A, Al Raqqad N, Zarei-Ghanavati M, et al; The role of keratoprostheses. Eye (Lond). 2018 Jan;32(1):7-8. doi: 10.1038/eye.2017.287. Epub 2017 Dec 1.
- Fernandez MM, Afshari NA; Endothelial Keratoplasty: From DLEK to DMEK. Middle East Afr J Ophthalmol. 2010 Jan;17(1):5-8. doi: 10.4103/0974-9233.61210.
- Price FW Jr, Price MO; Evolution of endothelial keratoplasty. Cornea. 2013 Nov;32 Suppl 1:S28-32. doi: 10.1097/ICO.0b013e3182a0a307.
Continue reading below
Article history
The information on this page is written and peer reviewed by qualified clinicians.
Next review due: 17 Aug 2028
22 Sept 2023 | Latest version

Ask, share, connect.
Browse discussions, ask questions, and share experiences across hundreds of health topics.

Feeling unwell?
Assess your symptoms online for free