Thyroid eye disease
Peer reviewed by Dr Hayley Willacy, FRCGP Last updated by Dr Colin Tidy, MRCGPLast updated 21 Apr 2023
Meets Patient’s editorial guidelines
- DownloadDownload
- Share
- Language
- Discussion
- Audio Version
Medical Professionals
Professional Reference articles are designed for health professionals to use. They are written by UK doctors and based on research evidence, UK and European Guidelines. You may find the Thyroid eye disease article more useful, or one of our other health articles.
In this article:
Synonyms: thyroid ophthalmopathy, dysthyroid eye disease, Graves' ophthalmopathy, ophthalmic Graves' disease, Basedow's disease (in German-speaking parts)
Thyroid eye disease (TED) is the most common extra-thyroidal manifestation of Graves' disease. It may also be associated with other forms of thyroid dysfunction and it may also occur in the absence of any biochemical thyroid dysfunction at all.
About 90% of patients with thyroid eye disease have Graves' hyperthyroidism, but hypothyroidism and euthyroidism can also accompany TED. Eye disease usually develops in patients with hyperthyroidism within the first 1-2 years, but the disease can occur decades later.1
Management is challenging and requires a team approach. Medical management is only available in the active, early phase. When the disease becomes inactive (no acute inflammation), surgery is the only option. TED is sight-threatening in up to 5% of patients, although most cases are mild and self-limiting.2 See also the separate articles Thyroid Function Tests, Hyperthyroidism, Hyperthyroidism in Pregnancy, Hyperthyroid Crisis (Thyrotoxic Storm), Hypothyroidism, Hashimoto's Thyroiditis and Eye in Systemic Disease.
Continue reading below
What is thyroid eye disease?3
TED is an organ-specific idiopathic autoimmune disease. It usually begins with an active inflammatory orbital phase lasting 6-24 months, during which expansion of extraocular muscles and orbital fat occurs.
This can result in proptosis, compressive optic neuropathy (CON) and compromised extraocular muscle motility. There is progressive deterioration initially, followed by a peak before spontaneously improving.
The inflammatory phase is followed by an 'inactive' fibrotic phase.4
The inactive phase is a chronic 'burn-out' phase where further changes are unlikely.
Sight loss can occur, usually when there has been delay in starting treatment.5
The onset of hyperthyroidism and TED are usually within 18 months of one another. However, TED may also develop years before or after the onset of hyperthyroidism. A small minority of patients never develop any associated thyroid disease. Most patients with TED have clinical and/or biochemical evidence of hyper/hypothyroidism but some are euthyroid (at least at the time of presentation). Therefore, thyroid dysfunction can precede, proceed or be co-existent with TED.
Pathogenesis6
Back to contentsThe pathogenesis of TED is still uncertain, but is thought to involve an autoimmune reaction to thyroid-stimulating hormone (TSH) receptors, modulated by T-cell lymphocytes.
Infiltration of lymphocytes into orbital tissue stimulates the release of cytokines (eg, tumour necrosis factor and interleukin-1) which promote the release of mucopolysaccharides from fibroblasts.The resulting hyperosmotic shift leads to oedema of the orbital fat and extraocular muscles, forcing the eyeball forward and leading to the typical appearance of exophthalmos. There are underlying genetic factors which are currently being researched.
Continue reading below
How common is thyroid eye disease? (Epidemiology)7 8
Back to contentsTED is the most common cause of orbital inflammation and proptosis in adults.9
TED affects an estimated 400,000 people in the UK, a prevalence of 0.7%.
About 90% of this is associated with Graves' disease.1
At least 50% of patients with Graves' disease develop clinically significant TED.10
10-15% of patients with TED have never been hyperthyroid and some are hypothyroid at the time the orbitopathy presents.
Risk factors2
Current smoking increases the risk of developing TED (relative risk of 7 for heavy smokers) although ex-smokers are not at increased risk.11 Risk increases with the number of cigarettes smoked and reduces on quitting.12 Smoking also increases the risk of ophthalmopathy after radio-iodine, although this can be reduced by corticosteroids.13 Smoking also delays and reduces the efficacy of the other methods of treatment such as steroids and radiotherapy.14
Female sex (due to the higher prevalence of thyroid disease in women).
Middle age.
There are some associated genes including HLA-DR3, HLA-B8 and the genes for CTLA4 and the TSH receptor.15
Autoimmune thyroid disease.
Uncontrolled thyroid dysfunction. Thyroid dysfunction is associated with more severe TED, and tight control of thyroid function appears to reduce the severity of TED.
Radio-iodine therapy is associated with progressive Graves' ophthalmopathy.13 16 Thus, it can only be used in the inactive phase of the eye disease (see 'Description', above).
Thyroid eye disease symptoms (presentation)3
Back to contentsMost patients present with concurrent thyrotoxicosis due to Graves' disease.
About 20% of patients develop eye problems in the months before becoming thyrotoxic.
About 10% present with current or previous hypothyroidism.
Occasionally, TED precedes thyroid dysfunction by several years.17
TED is the most common cause of proptosis in adults and should always be suspected in adults with unexplained diplopia, eye pain or optic nerve dysfunction.10
Ophthalmic features18
Thyrotoxicosis
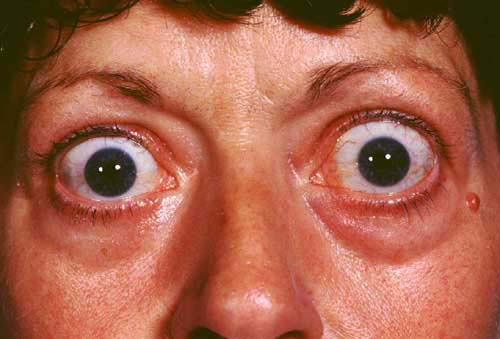
© Jonathan Trobe, M.D. - University of Michigan Kellogg Eye Center, CC BY 3.0, via Wikimedia Commons
The symptoms reflect the intensity of the inflammatory reaction and the severity of the anatomical, functional and cosmetic aspects. Initially, they relate to an increasingly 'crowded' orbit:
Ocular irritation.
Ache (worse in the mornings) behind the eye, especially when looking up, down or sideways.
Red eyes.
Cosmetic changes.
Diplopia (restricted ocular mobility, initially involving the inferior rectus muscles).
Change in the appearance of the eyes (usually the patient or family notices staring or bulging eyes).
Dry or watery eyes.
Mild photophobia.
Swelling of the upper and lower lids.
Redness of the lids and eyes.
Difficulty moving the eyes.
Proptosis (exophthalmos) may develop gradually, accompanied by:
Lid retraction and lid lag.
Conjunctival injection and chemosis (oedema).
Orbital fat prolapse.
Exposure keratopathy (photophobia, tearing, grittiness, pain) due to incomplete lid closure.
Exophthalmos does not always develop and it does not correlate with disease severity. Some patients with minimal exophthalmos are at high risk of optic nerve compression.19 Elderly patients may present with relatively inactive orbitopathy and progressive strabismus.19
Acute progressive optic neuropathy20
If the involved tissues (mainly muscle) begin to compress the optic nerve and if the nerve is further stretched due to proptosis, visual loss can occur. Prompt treatment offers a better chance of a good outcome.
Features suggesting optic neuropathy (and necessitating urgent referral) include:
Blurred vision.
Impaired colour perception.
Reduced visual acuity.
A relative afferent papillary defect.
Visual field defect.
See the separate Examination of the Eye article. See also below under urgent referral.
Systemic features
These depend on the thyroid status and the underlying disease. See the links at the beginning of this article for more information about dysthyroid states.
Continue reading below
Diagnosing thyroid eye disease21
Back to contentsThis is straightforward in patients with obvious bilateral eye disease and a background of thyroid function abnormalities. It can be more difficult in unilateral disease or when the patient is euthyroid.
Diagnosis is confirmed with blood biochemistry and orbital imaging - see 'Investigations', below.
Differential diagnosis21
Back to contentsTED is more often bilateral whereas non-TED proptosis (eg, from a retro-orbital tumour) is usually unilateral.
Allergic conjunctivitis is a common misdiagnosis when periorbital swelling and conjunctival injection are predominant: however, restricted eye movement, lid retraction ± blurred vision occur in TED and are not present in conjunctivitis.
Dry eye is a common misdiagnosis in early disease.
Orbital myositis.
Chronic progressive external ophthalmoplegia.
Idiopathic orbital inflammatory disease.
Lymphoproliferative disorders.
Caroticocavernous fistula.
Obesity can sometimes result in a similar clinical picture.23
Investigations21
Back to contentsTSH and free thyroxine (FT4). If normal but clinical suspicion remains, free tri-iodothyronine (T3).
Thyroid auto-antibodies: anti-TSH receptor, anti-thyroid peroxidase and anti-thyroglobulin antibodies (although these have poor sensitivity and specificity).
CT or (preferably) MRI of the orbits. MRI is better at showing soft tissue; CT is helpful if surgery for orbital decompression is planned. There will be enlarged extra-ocular muscles (with tendon sparing) ± an increase in orbital fibro-adipose tissue.
Thyroid uptake scan or orbital biopsy may be helpful.
Orthoptist review to fully assess ocular movement and visual fields.
Associated diseases3
Back to contentsTED is most commonly associated with Graves' disease: clinically apparent TED occurs in about 50% of cases of Graves' disease.
Thyroid eye disease may precede or follow endocrine manifestations but they tend to present within 18 months of each other in 80% of patients.
Even in the absence of clinical signs, imaging reveals subtle orbital changes in most cases of Graves' disease.
TED shows a significant association with autoimmune thyroiditis, such as in Hashimoto's thyroiditis, where it occurs in about 3% of cases.
Staging24
Back to contentsThere are various staging systems. The European Group on Graves' Orbitopathy severity assessment is as follows:
Mild thyroid eye disease
Minor impact on activities of daily living.
Insufficient justification for immunosuppression or surgical treatment.
One or more of the following:
Minor lid retraction (less than 2 mm).
Mild soft tissue involvement.
Proptosis less than 3 mm above normal for race and gender.
No or transient diplopia.
Corneal exposure responsive to lubricants.
Moderate-to-severe thyroid eye disease
Impact on activities of daily living.
Justification for immunosuppression and/or surgical treatment.
Two or more of the following:
Lid retraction 2 mm or more.
Moderate or severe soft tissue involvement.
Proptosis ≥3 mm above normal for race and gender.
Diplopia (inconstant or constant).
Sight-threatening thyroid eye disease
Compressive optic neuropathy.
Corneal ulceration.
Thyroid eye disease treatment and management2 6 25 26
Back to contentsNon-surgical management includes observation alone to therapy such as oral selenium, glucocorticoid therapy, ciclosporin, mycophenolate, rituximab, immunoglobulin, teprotumumab, and orbital radiotherapy. High-dose intravenous methylprednisolone therapy is used in active vision-threatening disease with early use of tarsorrhaphy and orbital decompression. Inactive but moderate to severe disease may be treated by orbital decompression, strabismus and eyelid surgery.24
General points
Ideally TED should be managed in a joint endocrinology and ophthalmology clinic where there is experience and expertise in the condition. The role or primary care professionals includes:
Identifying sight-threatening eye complications early if the patient is not already under specialist care and referring early (see 'Referral', below).
Supporting affected patients to stop smoking.
Achieving and maintaining euthyroid state.
Prescribing ocular lubricants where there are symptoms of corneal exposure.
Reminding patients to try sleeping propped up and to avoid dusty conditions.
Providing support group information (see 'Further reading & references', below).
Referral5
Patients with a history of Graves' disease, who have neither symptoms nor signs of TED, require no further ophthalmological assessments and need not be referred to a specialist. Where there is suspected TED, early referral is important because medical treatment is most effective when the eye is acutely inflamed.
Corneal exposure.
Strabismus.
Pressure on the optic nerve.
Poor cosmetic result.
EUGOGO also recommends urgent referral where:27
There is unexplained deterioration in vision.
Colour vision changes in one or both eyes.
There is sudden exophthalmos.
Corneal opacity is obvious.
There is restriction of eye movement.
There is disc swelling.
Treatment2 28
Treatment options include:
Prisms to control diplopia.
Botulinum toxin to reduce upper lid swelling.
Medical treatments:
Oral or systemic steroids.
Octreotide and lanreotide.
Ciclosporin.
Intravenous immunoglobulin.
Emerging treatments:
Selenium.
Rituximab.
Anti-tumour necrosis factor.
Orbital radiotherapy.
Surgical interventions:
Orbital decompression.
Strabismus surgery.
Lid-lengthening surgery.
Blepharoplasty.
Treatment of mild TED
Most patients have mild TED, with symptoms limited to dry eye and mild diplopia.
These are easily treated with artificial tears, ointment and prisms.
Conservative therapy has traditionally been recommended for these patients, although there is emerging evidence for early medical treatment.
Treatment of moderate-to-severe TED
Corticosteroids: the most commonly used medical therapy in active moderate-to-severe TED. It is not clear whether they alter the disease course. Studies suggest intravenous steroids are most effective.
Orbital radiotherapy now plays only a minor rule in the treatment of non-sight-threatening TED because studies and evidence reviews have not clearly demonstrated its efficacy.
Ciclosporin is less effective than corticosteroids but it may be of benefit in combination with oral corticosteroids as a steroid-sparing agent.
Rituximab is a monoclonal antibody that targets mature B cells. It is recommended in steroid-resistant patients but more evidence is needed before it replaces corticosteroids as a first-line treatment.
Surgical decompression is ultimately needed in up to 80% of patients with moderate-to-severe disease.
Treatment of sight-threatening TED
This accounts for around 5% of patients.
Urgent orbital decompression surgery and intravenous corticosteroid therapy both appear effective.
Prognosis
The natural history of the disease is variable: symptoms may progress, remain unchanged or improve spontaneously. Typically, the disease runs its course over a 12- to 24-month period.16
In thyrotoxic patients, 90% of proptosis improves and 30% of restrictive myopathy improves but proptosis rarely improves without further treatment. Poor prognostic factors include:29
Older age at onset.
Rapid progression at onset.
Longer duration of active disease.
Drop in visual acuity during the active phase.
Male gender.
Smoker.
Diabetes.
More than a third of patients are dissatisfied with the appearance of their eyes when the disease has run its course and more than a quarter have low visual acuity. Between about 10% and 35% will need further treatment.30
Dr Mary Lowth is an author or the original author of this leaflet.
Further reading and references
- Thyroid eye disease; Royal National Institute of Blind People (RNIB)
- British Thyroid Foundation
- EUGOGO (EUropean Group On Graves' Orbitopathy)
- Weiler DL; Thyroid eye disease: a review. Clin Exp Optom. 2017 Jan;100(1):20-25. doi: 10.1111/cxo.12472. Epub 2016 Oct 4.
- Neag EJ, Smith TJ; 2021 update on thyroid-associated ophthalmopathy. J Endocrinol Invest. 2022 Feb;45(2):235-259. doi: 10.1007/s40618-021-01663-9. Epub 2021 Aug 20.
- Belliveau MJ, Jordan DR; Thyroid eye disease. CMAJ. 2013 Jun 11;185(9):797. doi: 10.1503/cmaj.121815. Epub 2013 Apr 2.
- Gillespie EF, Smith TJ, Douglas RS; Thyroid eye disease: towards an evidence base for treatment in the 21st century. Curr Neurol Neurosci Rep. 2012 Jun;12(3):318-24. doi: 10.1007/s11910-012-0256-9.
- Weiler DL; Thyroid eye disease: a review. Clin Exp Optom. 2017 Jan;100(1):20-25. doi: 10.1111/cxo.12472. Epub 2016 Oct 4.
- Bothun ED, Scheurer RA, Harrison AR, et al; Update on thyroid eye disease and management. Clin Ophthalmol. 2009;3:543-51. Epub 2009 Oct 19.
- Perros P, Neoh C, Dickinson J; Thyroid eye disease. BMJ. 2009 Mar 6;338:b560. doi: 10.1136/bmj.b560.
- Yoon JS, Kikkawa DO; Thyroid eye disease: From pathogenesis to targeted therapies. Taiwan J Ophthalmol. 2022 Jan 21;12(1):3-11. doi: 10.4103/tjo.tjo_51_21. eCollection 2022 Jan-Mar.
- Lazarus JH; Epidemiology of Graves' orbitopathy (GO) and relationship with thyroid disease. Best Pract Res Clin Endocrinol Metab. 2012 Jun;26(3):273-9.
- Cawood T, Moriarty P, O'Shea D; Recent developments in thyroid eye disease: BMJ 2004; 329, Published 12 August 2004
- Zoumalan CI, Cockerham KP, Turbin RE, et al; Efficacy of corticosteroids and external beam radiation in the management of moderate to severe thyroid eye disease. J Neuroophthalmol. 2007 Sep;27(3):205-14.
- Cockerham KP, Chan SS; Thyroid eye disease. Neurol Clin. 2010 Aug;28(3):729-55. doi: 10.1016/j.ncl.2010.03.010.
- Bartalena L; Prevention of Graves' ophthalmopathy. Best Pract Res Clin Endocrinol Metab. 2012 Jun;26(3):371-9.
- Chan W, Wong GW, Fan DS, et al; Ophthalmopathy in childhood Graves' disease. Br J Ophthalmol. 2002 Jul;86(7):740-2.
- Ponto KA, Zang S, Kahaly GJ; The tale of radioiodine and Graves' orbitopathy. Thyroid. 2010 Jul;20(7):785-93.
- Krassas GE, Wiersinga W; Smoking and autoimmune thyroid disease: the plot thickens. Eur J Endocrinol. 2006 Jun;154(6):777-80.
- Ploski R, Szymanski K, Bednarczuk T; The genetic basis of graves' disease. Curr Genomics. 2011 Dec;12(8):542-63. doi: 10.2174/138920211798120772.
- Bartalena L, Tanda ML; Clinical practice. Graves' ophthalmopathy. N Engl J Med. 2009 Mar 5;360(10):994-1001.
- Maheshwari R, Weis E; Thyroid associated orbitopathy. Indian J Ophthalmol. 2012 Mar-Apr;60(2):87-93.
- Maheshwari R, Weis E. Thyroid associated orbitopathy. Indian Journal of Ophthalmology. 2012;60(2):87-93.
- Chong K; Thyroid Eye Disease: a Comprehensive Review, Medical Bulletin, The Federation of Medical Societies of Hong Kong, Vol 15, No 10. 2010.
- Behbehani R; Clinical approach to optic neuropathies. Clin Ophthalmol. 2007 Sep;1(3):233-46.
- Rashad R, Pinto R, Li E, et al; Thyroid Eye Disease. Life (Basel). 2022 Dec 12;12(12):2084. doi: 10.3390/life12122084.
- Findling JW, Raff H; Cushing's Syndrome: important issues in diagnosis and management. J Clin Endocrinol Metab. 2006 Oct;91(10):3746-53. Epub 2006 Jul 25.
- Smolders MH, Graniewski-Wijnands HS, Meinders AE, et al; Exophthalmos in obesity. Ophthalmic Res. 2004 Mar-Apr;36(2):78-81.
- Hall AJH, Topliss DJ; Medical and surgical treatment of thyroid eye disease. Intern Med J. 2022 Jan;52(1):14-20. doi: 10.1111/imj.15067.
- Bhatti MT, Dutton JJ; Thyroid eye disease: therapy in the active phase. J Neuroophthalmol. 2014 Jun;34(2):186-97. doi: 10.1097/WNO.0000000000000128.
- Men CJ, Kossler AL, Wester ST; Updates on the understanding and management of thyroid eye disease. Ther Adv Ophthalmol. 2021 Jun 30;13:25158414211027760. doi: 10.1177/25158414211027760. eCollection 2021 Jan-Dec.
- Bartalena L, Baldeschi L, Dickinson A, et al; Consensus statement of the European Group on Graves' orbitopathy (EUGOGO) on management of GO. Eur J Endocrinol. 2008 Mar;158(3):273-85.
- Bartalena L, Baldeschi L, Dickinson AJ, et al; Consensus statement of the European Group on Graves' orbitopathy (EUGOGO) on the Management of Graves' orbitopathy. Thyroid. 2008 Mar;18(3):333-46. doi: 10.1089/thy.2007.0315.
- De Hoog J, Stravers S, Kalmann R; Recession of the inferior rectus muscle in Graves' orbitopathy. Eye (Lond). 2010 Jun;24(6):1011-7. Epub 2009 Nov 13.
- Cawood T, Moriarty P, O'Shea D; Recent developments in thyroid eye disease. BMJ. 2004 Aug 14;329(7462):385-90.
Continue reading below
Article history
The information on this page is written and peer reviewed by qualified clinicians.
Next review due: 19 Apr 2028
21 Apr 2023 | Latest version

Ask, share, connect.
Browse discussions, ask questions, and share experiences across hundreds of health topics.

Feeling unwell?
Assess your symptoms online for free